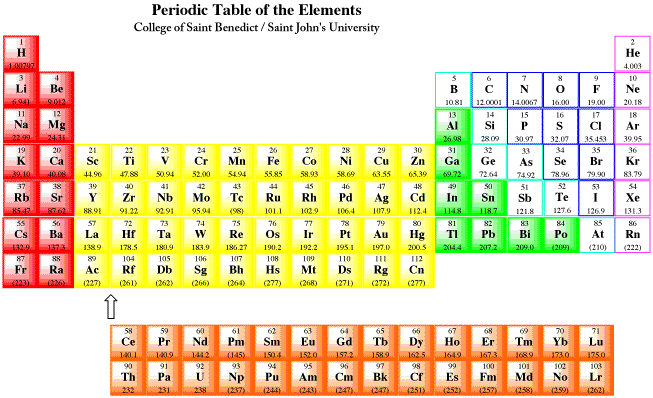
Reactivity series periodic table3/21/2024 Consider the partial activity series below to answer the questions. It is mostly based on tables provided by NIST. The following list includes the metallic elements of the first six periods. 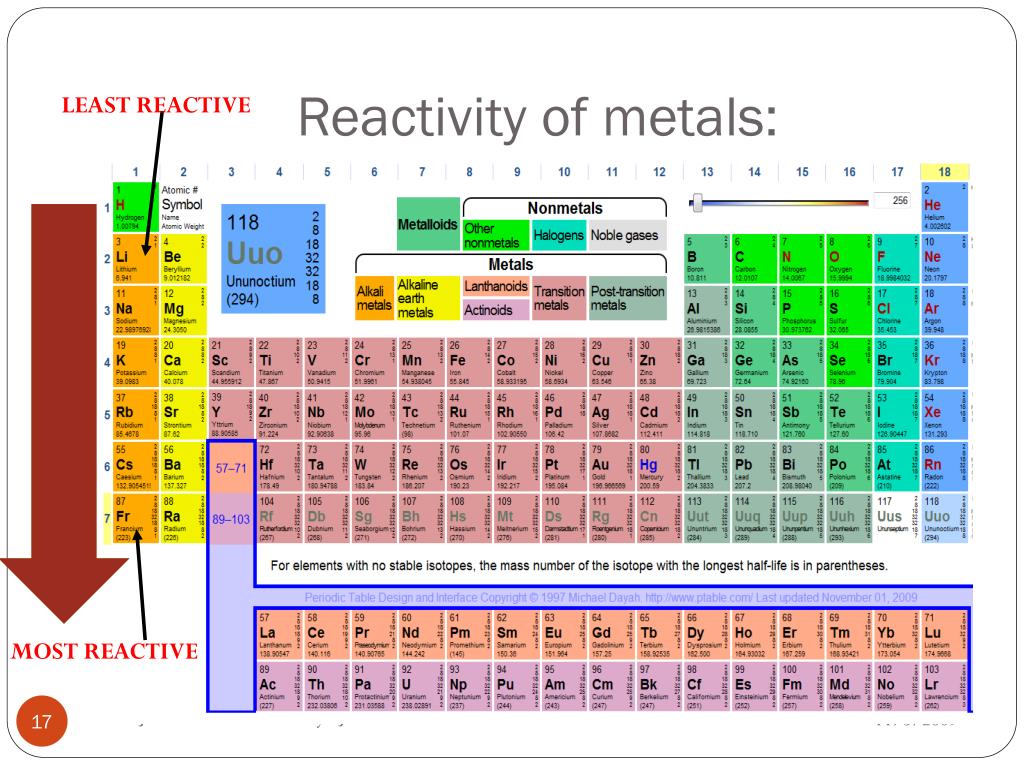
As we go from left to right on the periodic table, reactivity decreases.Įxercise 1. The reactivity series is sometimes quoted in the strict reverse order of standard electrode potentials, when it is also known as the 'electrochemical series'. They are much more stable meaning they are less reactive. The metals below hydrogen in the series do not react with aqueous hydrogen ions or water. The remaining metals (above hydrogen) will not react with water but do react with aqueous hydrogen ions. In fact, the first five metals in the series, both Group 1A and 2A metals, will react with pure water to form hydrogen gas.Ģ K (s) + 2 H 2O (l) c 2 K + (aq) + 2 OH – (aq) + H 2 (g) The elements are arranged in decreasing order of their reactivity. Reactivity series is an arrangement of metals and non-metals based on their tendencies to react with certain tendencies. The metals above hydrogen will react with aqueous hydrogen ion to form hydrogen gas. Reactivity is the tendency of chemical substances to form products by itself. Notice the position of hydrogen in the activity series. The aluminum is a stronger reducing agent than silver and will reduce the Ag + ion to solid Ag.ģ Ag 2S (s) + 2 Al(s) → 6 Ag (s) + Al 2S 3 (s) Tarnish can be removed chemically with aluminum. Silver tarnishes according to the following reaction with H 2S gas.Ģ Ag (s) + H 2S (gas) → Ag 2S (s) + H 2 (g)Ĭommercial silver polishes will remove the tarnish from silver, but some of the silver is lost because these polishes are abrasive. Chromium can reduce Cu 2+ ion, but it cannot reduce Mn 2+. A metal can reduce any ion below it in the series. For example, zinc is a weaker reducing agent than sodium, but it is a stronger reducing agent than copper. Gold is at the end of the list and is not as easily oxidized and is therefore the weakest reducing agent on this activity series.īoth the ease of oxidation and the reducing strength of the metals decrease going down the column. Lithium is at the top of the list and is the most easily oxidized metal which means it is the strongest reducing agent in the activity series below. 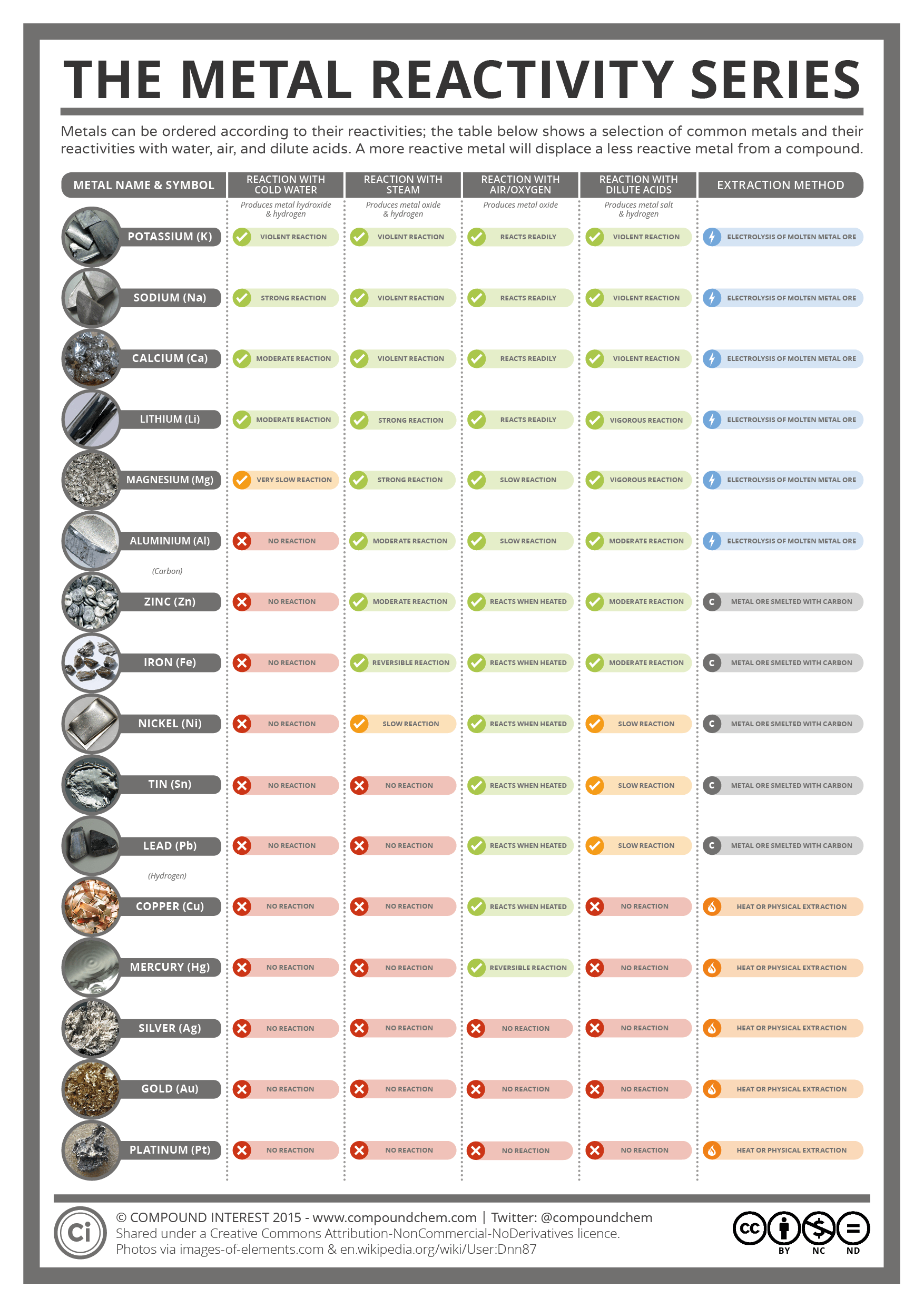
Below is a table of a partial activity series for metals in aqueous solution. Metals are strong reducing agents, but their reducing power decreases going across a period of the periodic table. Activity Series: An easy way to use the table is to note that a metal can reduce any ion of a metal below it in the table. This means metals lose electrons more easily than nonmetals. It is not possible.Metals are more easily oxidized than nonmetals. The activity series is a list of elements in decreasing order of their reactivity. There is no need to remember the entire series. In class XII it is used as the electrochemical series, a series of electrode potentials of different elements and ions. The Group 1 elements, also known as the alkali metals, all react vigorously with water to produce an alkaline solution. So the alkali metals in the bottom of the series is more reactive and non metals in the top of the series are more reactive. GCSE WJEC Group 1 alkali metals Explaining reactivity. In class XI it is used as the ability of the atom to donate or accept the electrons. As the reaction is in aqueous solution metals like Na, K are not considered in this series and only the series is Al>Zn>Fe>Cu etc. Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. In class X it is used as the reactivity of the metal to displace the another metal in aqueous solution. Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties. The word "reactivity series" is used in different ways in different classes.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
